Is TA-65 Anabolic?
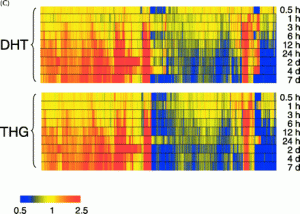
Testosterone causes altered gene expression in 939 genes as measured by a gene chip.
Human growth hormone causes altered gene expression in around five dozen genes.
In contrast, TA-65 appears to only slightly elevate genes in a a few areas related to DNA repair
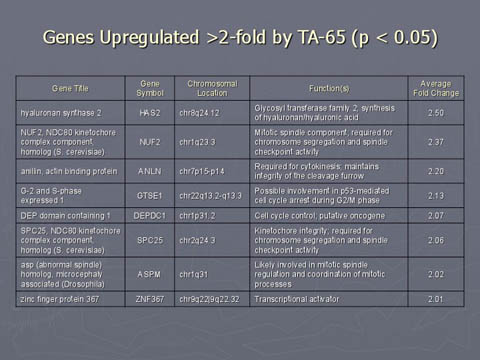












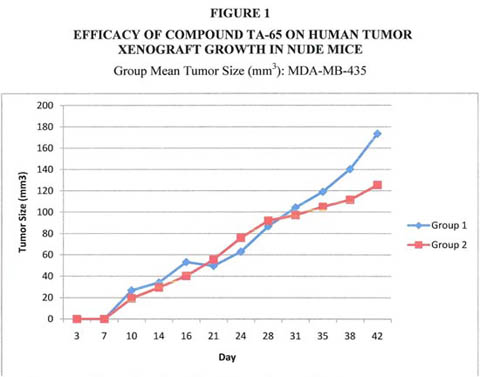 There was no statistical difference between TA-treated and placebo groups.
There was no statistical difference between TA-treated and placebo groups.