- Written:
- Author: Edward
- Posted in: dr ed park, research, stem cell theory of aging, TA-65, Telomerase Activation Medicine, Telomere erosion, testimonial
- Tags: Dr. Ed Park, halaschek-weiner, healthy aging, karl, short telomere, stem cells, super seniors, ta-65, telomerase activation, telomerase activation medicine, telomere
I would like to take you on ambitious journey. I will try to explain things that science has yet to validate but eventually (say 40-60 years), they will. SPOILER ALERT: good health cannot be measured with telomeres; it can only give an inference of current representation and doesn’t tell you how much pristine stem cell reserve you have left.
Let’s examine this landmark but ignored 2008 paper by Halaschek-Wiener et al [Mech Ageing Dev. 2008 Nov;129(11):638-41] entitled:
Reduced telomere length variation
in healthy oldest old.
“telomere length in 134 exceptionally healthy seniors aged 85 or older who have never been diagnosed with cancer, cardiovascular disease, major pulmonary disease, diabetes or Alzheimer disease (the ‘Super-seniors’) and 47 randomly-ascertained mid-life individuals aged 40-50 years. Super-seniors do not have exceptionally long telomeres for their age.
They have shorter telomeres than the younger control group. Variability in telomere length was lower in the Super-seniors than in the mid-life controls or the reference data. These results suggest that individuals, some types of their somatic cells, or both, may be selected for an optimal rather than extreme telomere length. “
MY CLINICAL OBSERVATIONS:
In fact, this paper reflects my best intuition about telomere measurements in my patients. As a result of the histograms provided by the LifeLength test (see below), I have noticed that after telomerase activation, we tend to see a disappearance of the extremely long and extremely short telomeres in the first two years of telomerase activation use which often results in a distressing and measurable SHORTENING of median length overall.
But there is usually stabilization and longer lengths with time. That is perhaps one of the issues that was not elucidated on the TA-65 randomized trial paper showing lengthening only in the one capsule group after one year.
If you are just going by median telomere length, you won’t get a reassuring picture in many older patients. That is because I believe some of the older hematopoetic stem cells have OVERGROWN telomeres from being senescent and that they undergo apoptosis in the first year of telomerase activation.
BASIC SCIENCE:
Imagine that when you were a fertilized egg, you had all 92 telomeres with 184 DNA strands all reset to a perfect 15,000 base pairs. Over time, some would shorten from damage, transcription error, or replicative senescence (getting old by copying). Over time, some would re-lengthen with telomerase.
The longer a stem cell lives, the more idiosyncrasies those 184 strands can accumulate. When you start to annihilate those damaged stem cells, the representation of telomere lengths reflects the younger, replacement cells’ telomeres.
Instead of hypothesizing that there is some better maintenance of telomere lengths, perhaps those super seniors just were lucky to have better stem cell apoptosis as well as high telomerase activity in their reserve stem cells to preserve genetic integrity. As always, disregard the last sentence of any abstract; here they say “selected” which assumes facts not in evidence.
CASE STUDY : Karl from age 72-77
If we take Karl’s telomeres as a case study, he started out with 7,000 median base pairs in 2011 by a Flow FSH assay by Repeat Diagnostics.
Imagine those telomeres were derived from some older stem cells which had acquired 72 years of variation from the original 15,000 length. Actually, they probably were somewhat ‘younger’ than 72 years because they may have been replaced more recently than fetal life.
After one year of taking TA-65, Karl’s median length dropped to 5,900 median as measured by a new company, Life Length. He was measured with 25% of the ends shorter than 3,000 base pairs.
These critically-short telomeres (<3kbp) are important because when they are too short, they cause mutation of the chromosomes by double strand DNA repair which makes double chromosomes and results in non-dysjunction mutation:
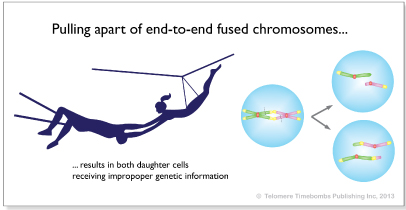
After 2 years, a 74yo Karl had even shorter median length of 4,900 base pairs with 27% critically-shortened. But he continued to feel great and improve so he stayed the course. This took courage because if he went by the telomere measurements alone and didn’t trust his body, he might not have had his future results.
WHY DID THE MEDIAN LENGTH DROP IN THE FIRST TWO YEARS
DESPITE FEELING BETTER?
If you induce apoptosis (cell death) in older, damaged stem cells, then you get a representation by younger or replacement stem cells with telomere lengths more closely distributed around the original middle of 15kbp for every telomere. (The original “middle” is merely an abstract concept. As you age, the best version of yourself has wild variation and generally shorter telomeres.)
With apoptosis and replacement, you don’t have those old and overgrown telomeres around as much. (This kind of drop in median length is not uncommon and something that no one is even discussing yet.)
After 3 years, a 75yo Karl had his best fantasy baseball week ever and his blood pressure continued to drop. He was now back up to 7,550 bp median length.
After 4 years, a 76yo Karl finally stabilized his median telomere length to 11,600 bp with around 3% critically-short telomeres.
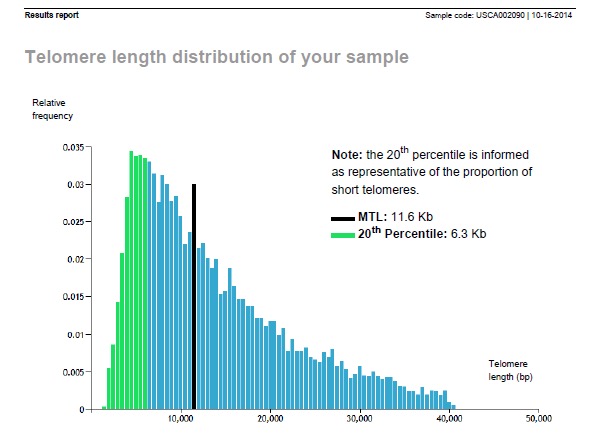
From there, he has stabilized with two subsequent measurements of 11,400 bp and 3% critically-short telomeres. and most recently, at age 77, 11,000 bp
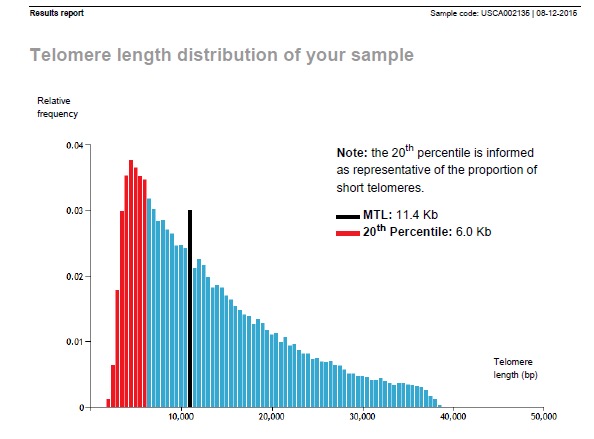
What is the take-home lesson?
In this one person, prolonged, high dose ingestion of TA-65 (4 capsules of 250U nightly) was associated with a 4,000 base pair (or a 40year equivalent) lengthening of telomeres that has stabilized and persisted over the last two years.
The critically-short, mutation-prone percentage of damaged telomeres has remained low at 3% after being as high as to 27%
Oh, and Karl is still having a great time and fantasy baseball camp and expanding his business ventures. We should all be as active, optimistic, and healthy as Karl.

