
General Disclaimer: This blog is for informational purposes only. It is not an advertisement for exosomes. Use of exosomes have not been FDA-approved for prevention nor treatment of any disease condition. Anecdotes provided do not constitute scientific proof and all patients were treated in the context of a fully informed consent and patient-physician relationship.
Exosomes 8: Disinformation or Bad Science?
Today, I had two interesting discussions: one was with a physician/entrepreneur who was promoting a private label exosome product and that led to a discussion with Dr. Duncan Ross of Kimera Labs, the makers of the exosomes that I am currently using.
The doctor I spoke with told me a rumor about the Kimera products stating that the product tested as containing only normal saline and therefore inert. She told me this was adversely affecting the reputation of Kimera. This doesn’t match my experience as I blogged about in my second blog of the series, so I called the CEO and chief scientist of Kimera, Dr. Ross, and he told me that he was fed up with having to address the issue.
The issue turns on something that I asked him about privately a month ago at his conference: if the exosomes are lipid bilayer spheres, how do you know what’s inside them? The scientist confessed that you need to first dissolve the membranes without destroying the contents. This is a non-trivial task involving a special mild detergent used in cell biology for just this purpose called Triton X-100 and protocols he developed specifically for this task which would not be used routinely if you just sent a vial to a lab and asked them to test it.
This statement of “nothing but saline” would be like sending a bag of toy balls suspended in saline and asking someone whether there were any toys. The answer would always be no unless you opened the balls, right? Why would someone want to get this misleading information out into the marketplace? It probably has something to do with competition and I can’t repeat Dr. Ross’s explanation of who and why this rumor was circulated.
Unless you gently dissolve the lipid bilayers of the exosomes with detergent that doesn’t also destroy the contents, you can’t know what’s inside. Actually testing a vial of purified exosomes (meaning no solutes from the stem cell broth included) should ALWAYS return a result of nothing but normal saline unless you let them degrade at room temperature to the point of membrane degradation. The whole point of the balls is to protect the proteins and RNA that are inside until they dock with your living cells!
These graphs showing the contents of Kimera exosomes may not be available from other suppliers unless they perform a similar detergent opening procedure.
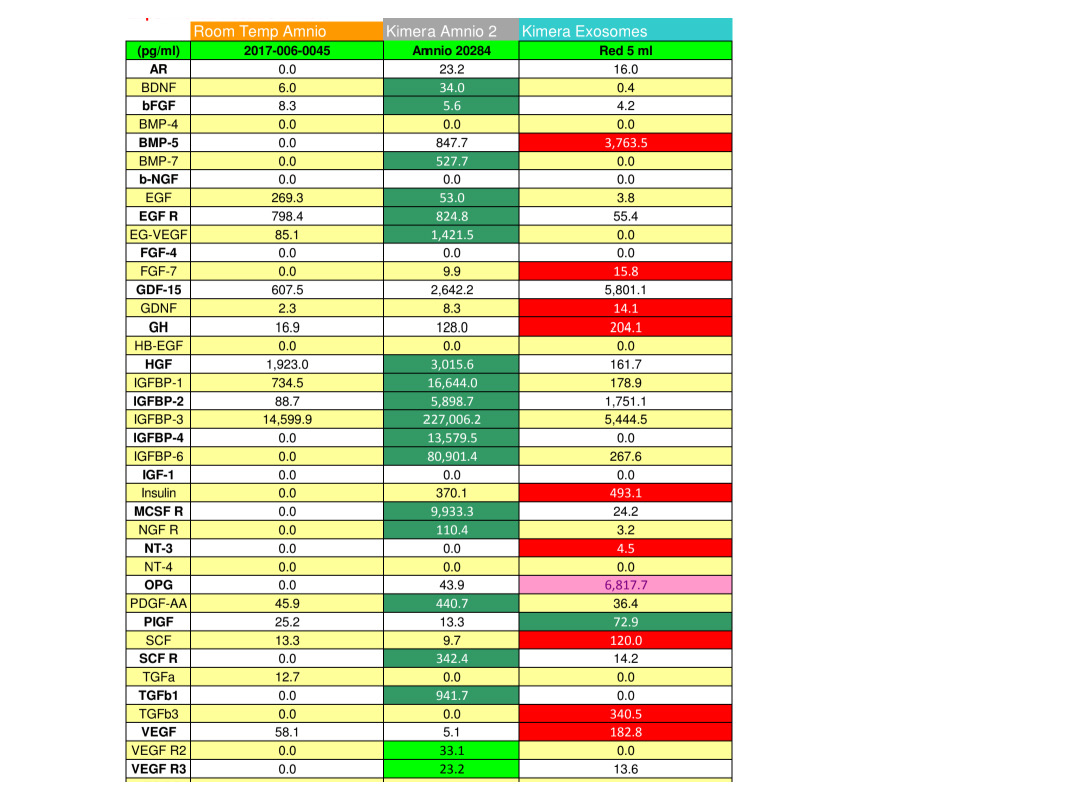
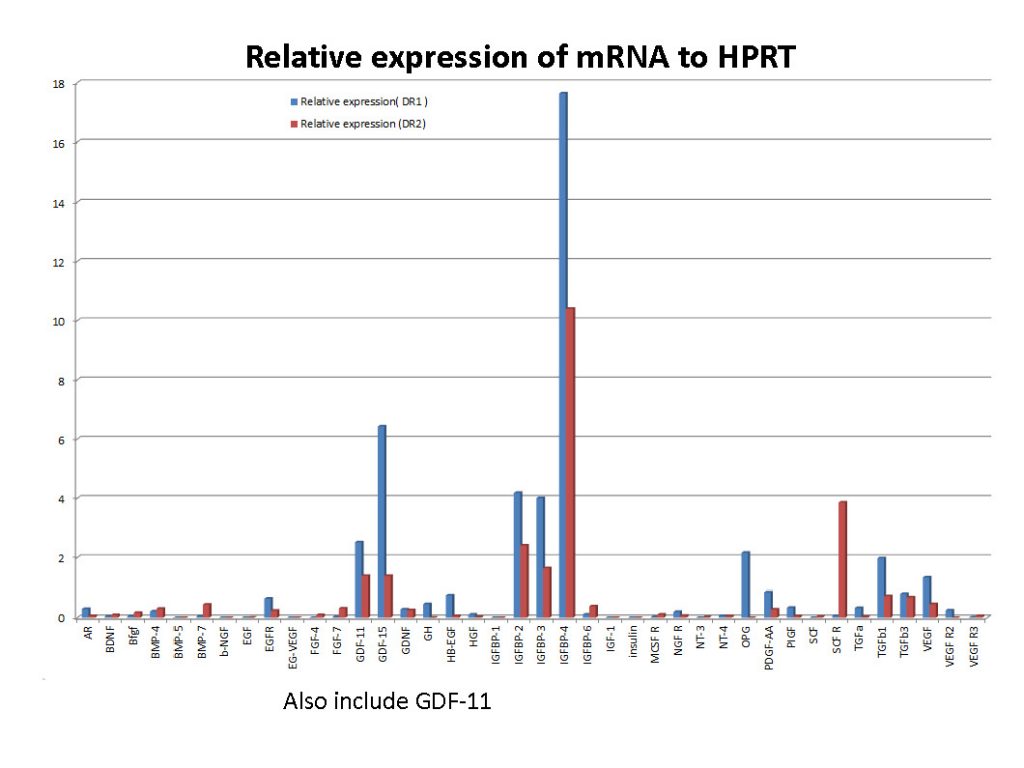
A machine known as a NanoSight study can only tell you the size and number of particles in suspension. Unless you do other things like examine them microscopically or segregate by surface proteins, you don’t know they’re the exosomes that we want.
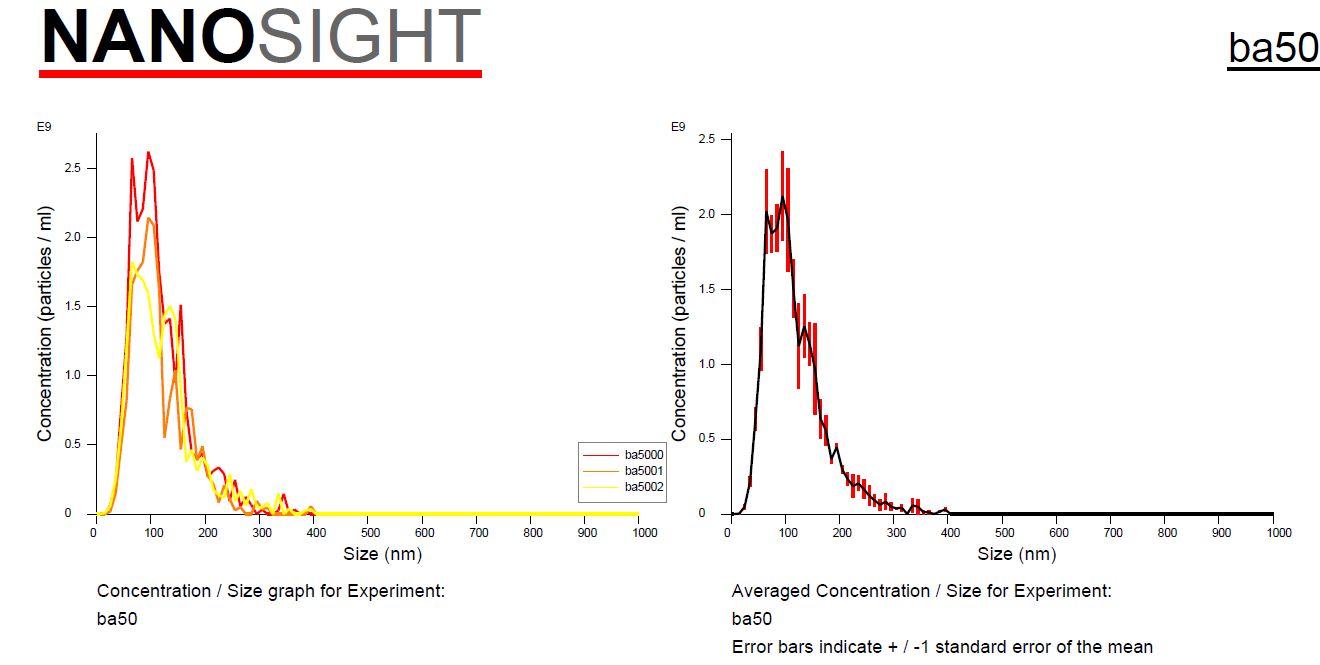
It cannot determine whether they are spherical exosomes with the correct surface markers that we want or merely debris such as pieces of meconium in the case of amniotic fluid extracted products. So ultimately we must rely upon the techniques and the integrity of the scientist at the labs to make sure they are extracting exosomes and not just inferring numbers from milligrams of assorted detritus that can be mixed in. When it comes to purchasing and using a product which require such expertise and integrity, it is important to have trust which is always earned by ongoing quality assurance. I can’t stress enough how important it is to know that a lab has integrity when it comes to producing this kind of acellular, resuspended, frozen product. I understand why Dr. Ross was upset but I hope this explanation clears up why testing a freshly thawed vial of exosomes should always return insignificant amounts of proteins and RNA. They are still inside the toy balls!
To understand more, please watch the 6th video in the nine-part series below. As a final aside, Dr. Ross said my best guesses as to how he makes his product was close but there are trade secrets that he uses to do an even better job of isolating only exosomes for his product. He seems to take a lot of pride in his craft, which arguably approaches an art when it comes to the variety of techniques available generally but that required him to invent others for internal use.
Feel free to bookmark the following link to have all my exosomes videos at your fingertips!
www.tinyurl.com/exosomesvideos
Archived exosome blogs….Please read them all.
N.B. On 2/29/2020 I changed my philosophy about keeping these blogs private. The archives up to blog 32 were appended to blogs that had yet to be created when the earlier ones were first written.
https://www.rechargebiomedical.com/exosomes-1-the-journey-begins/
I still have voluntary ASMR. Mom’s arthritis is greatly improved.
https://www.rechargebiomedical.com/exosomes-2-not-inert/
I explain why I believe exosomes aren’t inert. An amazing case of an overnight in heart rate variability is presented.
https://www.rechargebiomedical.com/exosomes-3-a-case-of-brain-and-neck-trauma/
A case of whiplash and traumatic brain injury improved after exosome use.
https://www.rechargebiomedical.com/exosomes-4-back-on-the-treadmill-again/
After exosome injection, two runners were able to return to running after limitations from tendinosis that plagued them for months (in the case of the 60-yo man, and years (in the case of the 53-yo woman)
https://www.rechargebiomedical.com/exosomes-5-clinical-medicine-is-bittersweet/
In this blog, I discussed how complicated clinical medicine can be. Different treatment protocols, ambiguous results ethical dilemmas and strange phenomena like acquired coffee aversion are part of what make treating with exosomes so challenging.
https://www.rechargebiomedical.com/exosomes-blog-6-healing-takes-time/
Using my left foot as an example, I attempt to explain that while some exosome effects are immediate, others take time. Remodeling of damaged tissues is a complicated process. My foot is still scarred and probably will be for life.
https://www.rechargebiomedical.com/exosomes-blog-7-nerve-pain-gone/
A patient after dental exosome injection becomes pain free after 4 years of suffering.
http://www.rechargebiomedical.com/exosomes-blog-8-disinformation-or-bad-science/
I explain why lab testing of freshly thawed exosomes is SUPPOSED to come back as normal saline.
https://www.rechargebiomedical.com/exosomes-blog-9-watch-all-my-exosome-videos/
A nine-part comprehensive explanation of exosomes.
https://www.rechargebiomedical.com/interviews-with-exosome-experts-posted/
I interviewed the principal scientists of Kimera Labs and other experienced clinicians using exosomes
https://www.rechargebiomedical.com/exosomes-blog-11-deb-bs-knees-are-improving
66-yo Deb B went from not being able to do one squat to 100 per day after exosome injection.
https://www.rechargebiomedical.com/80-yo-doug-ran-121-miles-in-6-days/
80-yo Doug describes in his video how much his knees have improved since getting exosome injections. He is reaching new athletic peaks with no signs of stopping.
https://www.rechargebiomedical.com/exosome-blog-13-microneedling-my-mom-with-exosomes/
My 82-yo mom underwent microneedling with exosomes with fantastic results. Most impressive was the absence of “downtime” from redness or bruising.
https://www.rechargebiomedical.com/exosome-blog-14-the-shoulder/
54-yo weightlifter with chronic pain from a rotator cuff tear shows remarkable improvement in just four days! He re-injured it at the gym so had to undergo a repeat shoulder injection
https://www.rechargebiomedical.com/exosomes-blog-15-face-blindness/
48-yo woman with severe face blindness experiences some improvement after nasal injections of exosomes. Her ability to remember new faces has improved permanently
https://www.rechargebiomedical.com/exosomes-blog-16-lecture-in-hawaii/
I delivered two lectures in Hawaii. The first was about exosomes generally and the second is a password-protected video about clinical applications.
https://www.rechargebiomedical.com/exosomes-blog-17-reading-is-fundamental
Three cases of middle-aged women reporting enhanced enjoyment and renewed capacity to read after nasal exosome therapy
https://www.rechargebiomedical.com/exosomes-blog-18-dental-health-improved
Two cases of improved dental health presented. We discuss the hazards of EMF and the promising future of dental regenerative therapy
https://www.rechargebiomedical.com/exosomes-19-chakra-balancing-with-exosomes/
We discuss the notion of energy centers known as chakras and the fact that we are electrical beings
https://www.rechargebiomedical.com/exosomes-blog-20-seborrheoic-dermatitis/
A case of improvement in Seborrheoic Dermatitis after microneedling with MSC exosomes is presented.
https://www.rechargebiomedical.com/exosomes-blog-21-bias-is-unavoidable/
I explain how poor we are at assessing change and how attribution and recall bias play important roles
https://www.rechargebiomedical.com/exosomes-22-badpress/
In the past year, there has been a lot of shifting loyalties in the exosome space resulting in a lot of rumor, innuendo, and bad press. With various parties trying to get each other in trouble with federal regulators, it is a treacherous business to be in.
https://www.rechargebiomedical.com/exosomes-blog-23-stasis-dermatitis
I describe a case of an 86-yo woman whose chronic leg rash disappeared after improvement in her leg circulation
https://www.rechargebiomedical.com/exosomes-blog-24-how-long-do-exosomes-last
I use a car wash analogy to answer the question “how long do exosomes last?” The answer is it depends on how dirty and damaged the car is, how well you clean it, and how dirty you get it after washing.
https://www.rechargebiomedical.com/exosomes-blog-25-knee-treatments/
I describe four cases of improvement of osteoartritic knees from my trip to Hawaii.
https://www.rechargebiomedical.com/exosomes-blog-26-the-placebo-effect/
I explain the three definitions of placebo and why I believe most of the effects of exosomes cannot be attributed to the so-called placebo effect.
https://www.rechargebiomedical.com/exosomes-27-soft-signs-of-exosome-actions/
Some remarkable “soft signs” of exosome effects include itching (new nerves), twitching (new muscles), and heaviness (a healing FORCE).
https://www.rechargebiomedical.com/exosomes-28-varicose-veins/
Three amigos came to see me and all enjoyed benefits. The best was the 48-yo gentleman who had a dramatic improvements to his irritable bowel syndrome, varicose veins, and appearance.
https://www.rechargebiomedical.com/exosomes-29-are-exosomes-smart/
I address the common observation from patients that exosomes seem “smart” or that they know where to go. In fact, I doubt this is true and that they are no smarter than player piano scrolls are good musicians.
https://www.rechargebiomedical.com/exosomes-30-real-exosomes-patients/
I gave a lecture at the University of Hawaii with about 80 attendees. Around 10 of them were actual patients. Hear in their own words how exosomes helped them.
https://www.rechargebiomedical.com/exosomes-blog-31-leg-swelling/
I describe two cases of improved leg swelling and explain what causes this common condition and how exosomes may be improving it.
https://www.rechargebiomedical.com/exosomes-32-nerve-regeneration
We present an amazing case of overnight nerve regeneration after ultrasound-targeted exosome injections.


7 thoughts on “Exosomes 8: Disinformation or Bad Science?”
Excellent explanation!
KUDOS!
YOU HAVE LITERALLY TAKEN THE SUPPORT OF A COLLEAGUE (and product) TO A WHOLE NEW SCIENTIFIC LEVEL….. I WISH OUR POLITICIANS DEALT WITH THE SAME TRUTH AND CLARITY…
ALOHA, MARIANNE WASSEL
Pingback: Exosomes 2: Not inert | Recharge Biomedical
Pingback: Exosomes 5: Clinical Medicine is Bittersweet | Recharge Biomedical
Pingback: Exosomes 34: Double vision | Recharge Biomedical
Pingback: Exosomes 36: Tinnitus | Recharge Biomedical
Pingback: Exosomes 39: The delayed reactions – Recharge Biomedical